
 Recent shifts in the competitive landscape have Mackie Research Capital analyst André Uddin raising his price target on Bellus Health (Bellus Health News, Stock Quote, Chart TSX:BLU).
Recent shifts in the competitive landscape have Mackie Research Capital analyst André Uddin raising his price target on Bellus Health (Bellus Health News, Stock Quote, Chart TSX:BLU).
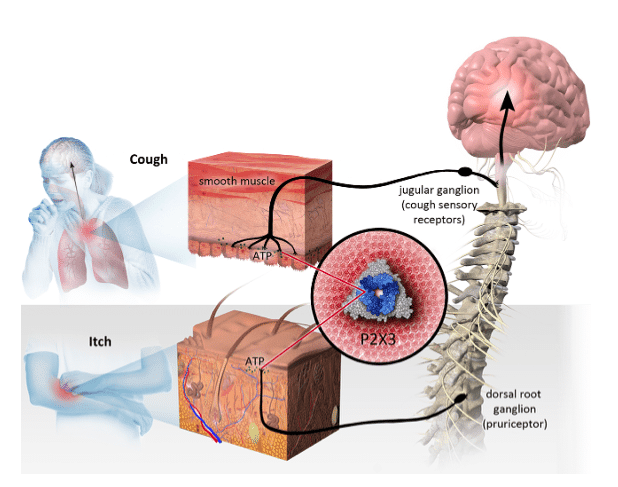
Uddin says in the landscape of chronic cough treatments, P2X3 inhibitors are showing the most promise and advanced class.
“Four classes of treatments have been tested for chronic cough – P2X3 inhibitors, NK1 antagonists, TRP channel modulators and α7 nAChR agonists,” the analyst says. “Two P2X3 inhibitors, Merck’s gefapixant (now in Phase III trials) and Shionogi’s S-600918 (now in Phase II trials), led to a significant >30% placebo-adjusted reduction in cough frequency in Phase II trials. Two NK1 antagonists (NeRRe’s orvepitant & Menlo’s serlopitant) failed to significantly improve cough reductions in Phase IIb trials. Two TRP channel modulators, GSK’s GSK2798745 (Phase II) and Alveonix’s AX-8 (pre-clinical), were discontinued due to efficacy futility. Attenua’s α7 nAChR agonist failed in its Phase II trials.”
Uddin says Bellus is transforming into a P2X3 platform company, a move that should bode well in the compettitive landscape.
“BLU-5937’s higher selectivity toward P2X3 should set apart the company from other P2X3 players,” he adds. “The KOL meeting highlighted potential broad applications of P2X3 inhibitors. Merck’s recent analyst day meeting showcased the same – potential indications include endometrial-related pain (now in Phase II trials), sleep apnea and sensory function-related disorders – each represents a significant market opportunity. Bellus noted it is developing BLU-5937 in pre-clinical studies for a new indication (undisclosed) – BLU-5937 would enter a Phase II trial directly for this indication.”
In a research update to clients today, Uddin maintained his “Speculative Buy” rating but raised his one-year price target on Bellus Health from $1.90 to $4.10, implying a return of 39 per cent at the time of publication.
Uddin thinks BLU will post and EPS loss of $0.14 on zero revenue in fiscal 2019. He expects those numbers will improve to an EPS loss of $0.09 on revenue of $3.8-million the following year.
Leave a Reply
You must be logged in to post a comment.





 Share
Share Tweet
Tweet Share
Share




Comment