Bellus Health has a 47 per cent upside, Mackie Research says


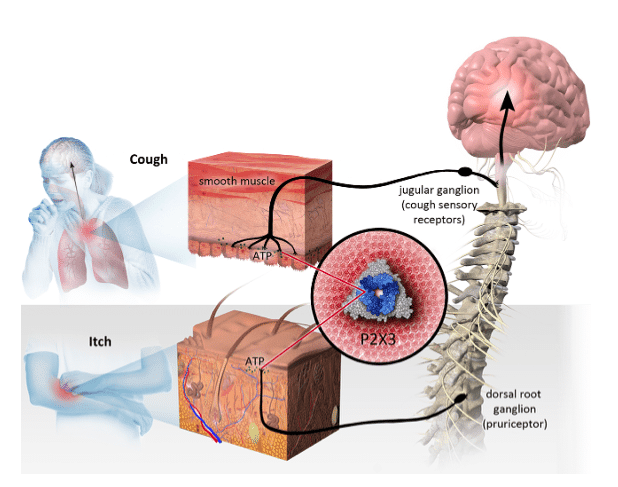
Bellus Health (Bellus Health Stock Quote, Chart TSX:BLU) is all cashed-up to start a Phase II trial of its chronic cough drug, one which analyst André Uddin of Mackie Research calls a potentially best in class opportunity to target a multi-billion dollar market.
Yesterday, clinical-stage drug company Bellus Health announced that it had closed on a $35 million equity financing round at $0.95 per share (36.8 million shares), which the company will put towards a Phase II study of its chronic cough drug BLU-5937, said to commence by mid-2019 with results available by mid-2020.
Uddin says that Bellus could be presented with a decent monetization opportunity after the Phase II trial.
“We believe the Phase I data of BLU- 5937, together with the potential success of the Phase II study, should help Bellus forge a licensing deal for this candidate (Bellus could also be acquired),” Uddin says in a client update on Tuesday. “To be conservative, we have assumed Bellus would out-license BLU-5937 in 2020 (post Phase II) for an upfront of C$150 million and contingent milestones of C$450 million, as well as a royalty of 20 per cent in upfront.”
For comparison, Uddin points to Merck’s 2016 acquisition of Afferent Pharmaceuticals for its clinical trial stage cough drug MK-7264, which Merck paid US$500M upfront.
Uddin’s estimates have BLU generating revenue in 2020, 2021 and 2022 of $3.8 million, $7.5 million and $82.5 million, respectively.
The analyst has reiterated his “Speculative Buy” rating but has reduced his target price from $2.20 to $1.90 due to share dilution. The $1.90 target represents a projected return of 47 per cent at the time of publication.