Oncolytics Biotech and Orphan Drug Designations: Wash, Rinse, Repeat
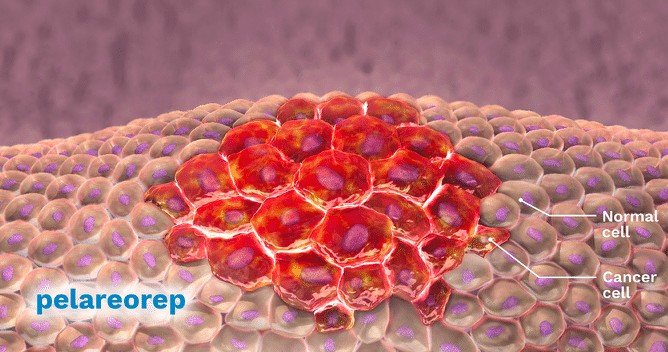
 Oncolytics Biotech (Oncolytics Biotech Stock Quote, Chart, News: TSX:ONC, Nasdaq:ONCY) yesterday announced it had received orphan drug designation from FDA for their oncology drug, REOLYSIN, in the treatment of primary peritoneal cancer. This is the fourth time in less than a month that the company has announced an FDA orphan drug designation for REOLYSIN. Prior to yesterday’s orphan drug designation announcement for peritoneal cancer, Oncolytics had issued almost verbatim news for ovarian, fallopian tube, and pancreatic cancer indications. In the time between announcing their first and fourth orphan designation Oncolytics stock has more than doubled.
Oncolytics Biotech (Oncolytics Biotech Stock Quote, Chart, News: TSX:ONC, Nasdaq:ONCY) yesterday announced it had received orphan drug designation from FDA for their oncology drug, REOLYSIN, in the treatment of primary peritoneal cancer. This is the fourth time in less than a month that the company has announced an FDA orphan drug designation for REOLYSIN. Prior to yesterday’s orphan drug designation announcement for peritoneal cancer, Oncolytics had issued almost verbatim news for ovarian, fallopian tube, and pancreatic cancer indications. In the time between announcing their first and fourth orphan designation Oncolytics stock has more than doubled.
The FDA can grant orphan drug designation for drugs targeting diseases affecting fewer than 200,000 people in the U.S. Having the orphan designation offers Oncolytics both potential pre-approval and post-approval benefits, including tax credits, reduction or elimination of certain FDA administration fees, and most importantly seven years market exclusivity if REOLYSIN were to be approved.
Investors tend to overreact to orphan designations, perhaps incorrectly correlating them with the likelihood of clinical success for the designated compound. True, an orphan designation could be very powerful, if REOLYSIN were to ever to be FDA approved for one of these four rare cancer indications. But the data thus far for REOLYSIN, in these orphan indications, are ambiguous at best.
As an example, in September 2014, the company announced Phase II data in pancreatic cancer, an indication where they have subsequently received an FDA orphan designation, that showed minimal survival benefit by adding REOLYSIN to standard of care. Oncolytics did share additional data from the study suggesting a sub-group of pancreatic cancer patients experienced greater survival benefit with REOLYSIN treatment, but it wasn’t able to convince the market, as the stock took a 50% nose dive in the days following the Phase II study results announcement.
The market may not love Oncolytics’ clinical data, but it does seem to love the plethora of orphan drug designation press releases the company has been able to generate of late. In fact the stock, on the back of the four orphan drug designation announcements, has returned to the same level it was before it’s disappointing Phase II results in pancreatic cancer.
Fortunately, investors will likely get more of what they like in the future, as Oncolytics has publicly stated that it has filed for FDA orphan drug designation for Reolysin in the treatment of pediatric patients with high grade gliomas and with the European Medicines Agency for orphan drug designation for Reolysin for the treatment of pancreatic and ovarian cancer. Oncolytics and orphan drug designations: wash, rinse, repeat……

Hogan Mullally
Writer