5 Questions with Thomas Braun of Verisante (TSXV:VRS)
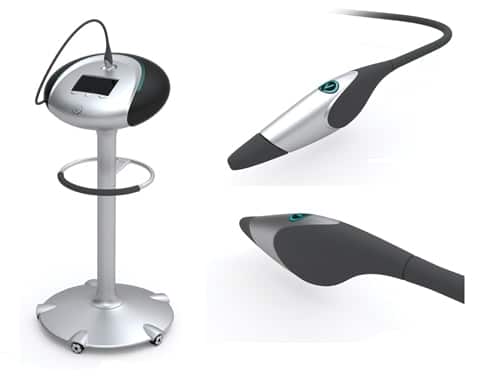
Imagine a hospital without an MRI machine. Or a medical centre that couldn’t take an x-ray. If Verisante’s (TSXV:VRS) vision of the future plays out, and some very smart people are betting it will, the idea of a medical professional without a Verisante Aura skin cancer detection device or a Verisante Core for detection of other cancers will seem just as foolhardy.
Founded in 2006 in Vancouver, Verisante Technology designs medical devices for the early detection of cancer. The Company’s initial focus is skin cancer, including basal cell carcinoma, squamous cell carcinoma, and melanoma. Verisante has licensed a technology developed by the BC Cancer Agency for in vivo, real-time, non-invasive skin measurements. This platform technology may also be applied to early detection systems for other types of cancer, including lung, GI, colorectal, and cervical cancers. The company expects Health Canada approval for both devices later this year.
Cantech Letter talked to Verisante CEO Thomas Braun.
Tell us a bit about your business. How do you make money?
Verisante has two product lines in the pipeline: Verisante Aura(TM) for skin cancer detection and the Verisante Core(TM) series for lung, colon and cervical cancer detection. Each of these product lines utilize the same proprietary cancer detection platform, while the operating software and probe technology are unique to each device. The cancer detection platform was developed by the BC Cancer Agency and tested and refined on approximately 1,000 lesions at the Skin Care Centre at Vancouver General Hospital. This exclusive platform technology allows Verisante to develop and offer a range of compact, non-invasive cancer detection devices that offer physicians immediate results for many of the most common cancers.
__________________________________________________________________________________________________________________
Want the latest on the hottest Canadian tech stocks? Sign up for CANTECH BUZZ, our weekly newsletter. Simply enter your email in the space to the right..
_____________________________________________________________________________________________________________________
The Aura(TM) and Core(TM) series have not yet been approved for sale. The Company anticipates Health Canada approval for the Aura(TM) in late 2011. Once approved, the Company expects to employ a razor/razor blade revenue model, offering disposable end caps (necessary for sanitary reasons) for each of the devices.
Is your space growing? How are you positioned to benefit?
Skin cancer is the most common form of cancer and is growing at a faster rate than any other cancer. 1 in 5 Americans will get skin cancer in their life time. Melanoma is the #1 cancer killer of women aged 25-30. Verisante is addressing an unserviced market for skin cancer detection. The American Cancer Society advises everyone to get screened for skin cancer starting at age 20.
What is the most common misconception about your company or its business?
That it is prohibitively expensive and difficult to obtain regulatory approval because people confuse medical devices with pharmaceutical under the label of “biotech” or because they invested in an invasive medical device like a stent or heart valve. Our device is a Class 2 device in Canada and Europe and does not require Phase 1,2,3 like drugs, or lengthy expensive trials like implantable devices. A clinical trial for our screening device is simple and is relatively inexpensive because all we are doing is scanning a skin lesion in under a second prior to the biopsy. In our case a six year study at Vancouver General Hospital is already complete.
What’s your financial situation like?
Verisante has approximately $5 million in the bank which will carry us through to the full commercialization of the Aura for skin cancer in Canada, Australia and Europe.
What is your key competitive advantage?
Our device has very high sensitivity, specificity, works in real time and uses a tiny probe. It can be used by a doctor’s assistant, as it does not require specialized knowledge to interpret results.
Disclaimer: Verisante is a sponsor of Cantech Letter.
_______________________________________________________________________________________
_______________________________________________________________________________________
Nick Waddell
Founder of Cantech Letter
Cantech Letter founder and editor Nick Waddell has lived in five Canadian provinces and is proud of his country's often overlooked contributions to the world of science and technology. Waddell takes a regular shift on the Canadian media circuit, making appearances on CTV, CBC and BNN, and contributing to publications such as Canadian Business and Business Insider.