Medicenna Therapeutics has a huge upside, says Mackie Research
 In an update to clients on Thursday, Mackie Research analyst André Uddin reports that a potential competitor drug to Medicenna Therapeutics’ (Medicenna Therapeutics Stock Quote, Chart, News TSX:MDNA) lead orphan drug candidate MDNA55 has failed in a Phase III trial and that investors should be on the lookout for Medicenna’s upcoming presentation on its ongoing Phase IIb trial for MDNA55.
In an update to clients on Thursday, Mackie Research analyst André Uddin reports that a potential competitor drug to Medicenna Therapeutics’ (Medicenna Therapeutics Stock Quote, Chart, News TSX:MDNA) lead orphan drug candidate MDNA55 has failed in a Phase III trial and that investors should be on the lookout for Medicenna’s upcoming presentation on its ongoing Phase IIb trial for MDNA55.
Shares in cancer-selective gene therapy company Tocagen took a nosedive on Thursday on news of the failed test from Tocagen’s combination therapy candidate Toca 511/Toca FC in patients with recurrent high grade glioma (HGG), a form of brain cancer that includes glioblastoma. The drug candidate missed the primary endpoint of overall survival compared to standard of care treatment (chemotherapy or Avastin) — 11.1 months versus 12.2 months.
“Recurrent brain cancer represents a very high unmet medical need and patients with this disease have very few treatment options,” said Marty Duvall, CEO of Tocagen, in a press release. “While the Toca 5 trial results are disappointing, we will be conducting a thorough analysis of the data including molecular analyses and pre-planned subgroups.”
The result pulls a competitor off the table for Medicenna and its MDNA55 in development for glioblastoma, with Medicenna’s share price spiking over 20 per cent in Thursday trading.
Uddin is staying bullish on MDNA.
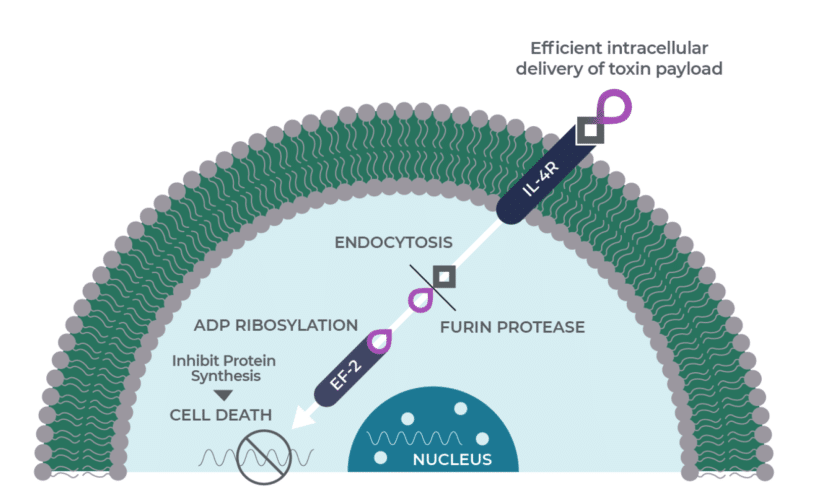
“Two doses of MDNA’s MDNA55 are being tested in a Phase 2b trial in recurrent GBM (rGBM) patients. Previously disclosed interim data from this trial showed the subjects on the low- dose MDNA55 cohort had an average overall survival of 11.8 months – 12.6 months in MDNA55 responders,” writes Uddin.
“Investors should realize that HGG is a less severe form of brain cancer than GBM and that MDNA55 is being studied in patients with the most severe form of brain cancer as they have failed nearly every previous line of treatment – this population represents arguably the most difficult-to-treat brain cancer patients,” he says.
Coming up on September 24 and 25, Medicenna will be presenting updated results on the on-going Phase IIb trial with MDNA55 at the Inaugural Targeting Innate Immunity Congress in Cambridge, Mass.
“We also expect MDNA to have an end-of-Phase II meeting with the FDA in late 2019 – from which the company should find out if results of the Phase 2b trial (assuming positive) are sufficient to support a BLA filing,” Uddin writes.
On top of those events, Uddin points to the initiation of a Phase II trial with MDNA55 in newly diagnosed GBM patients, expected by the end of 2019.
With the update, Uddin is maintaining his “Speculative Buy” rating and $3.30 target price, which represented a projected 12-month return of 244 per cent at the time of publication.
Uddin contends that his target price doesn’t include potential monetization opportunities of MDNA’s pre-clinical assets, which if deals do come would represent “significant upside potential” to MDNA’s current market cap and target price.
Earlier this year on June 18, Medicenna reported what Uddin calls promising interim top-line results for MDNA55 which not only demonstrated decent dose-dependent responses but also suggested that the drug candidate should have a high probability of meeting the trial’s primary endpoint of objective response rates.
Nick Waddell
Founder of Cantech Letter
Cantech Letter founder and editor Nick Waddell has lived in five Canadian provinces and is proud of his country's often overlooked contributions to the world of science and technology. Waddell takes a regular shift on the Canadian media circuit, making appearances on CTV, CBC and BNN, and contributing to publications such as Canadian Business and Business Insider.