Byron Capital: AEterna Zentaris is a Speculative Buy, Target Price $3.50

By the end of that month, the Quebec-based oncology/endocrinology drug maker had completed recruiting 465 patients for its perifosine phase 3 trial for refractory advanced colorectal cancer. And by October, two articles reporting positive phase one and two results perifosine had been published in the October, 2011 on-line issue of the Journal of Clinical Oncology.
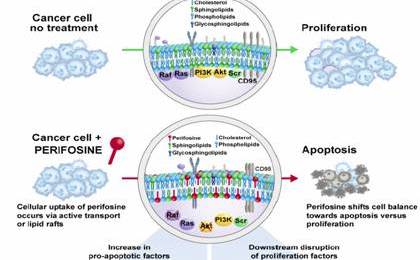
AEterna Zentaris has a lot riding on perifosine, a drug with multiple niche applications in cancer treatment. While the company has received encouraging news in the forms of FDA fast tracking and orphan status (for the treatment of neuroblastoma) some investors may have mistakenly believed the treatment itself only has niche potential, after all The FDA’s orphan drug status is reserved for new treatments that are being developed for diseases or conditions that affect fewer than 200,000 people in the United States. But the sum total of all opportunities for perifosine is quite large. According to Global Industry Analysts, the global market for Multiple Myeloma Therapeutics is forecast to reach US$6.4 billion by the year 2017.
In 2010, AEZ managed to stem its losses to $23.2 million from the nearly $60 million it lost in 2008, but was clearly in a race against time. Much of AEterna Zentaris’ recent revenues are are non periodic milestone payments for Cetrotide, an in vitro fertilization drug that is currently the company’s only marketed product.
While the red ink continued to pour this year; the company lost $10.05 million in this year’s Q1 and a further $10.56 in Q2, the stock began to revert to its old ways. Share of AEZ on the TSX (the company is also listed on the Nasdaq) fell from a high of $2.51 on June 1st to a recent low of $1.47 on October 14th.
But is a turnaround in the making or AEterna Zentaris? The company’s recently reported Q3 broke the string of heavy losses with a $1.07 million profit.
Byron Capital’s Healthcare and Biotechnology Analyst Douglas Loe says that while much of the value proposition he sees in the company still remains in its more advanced offerings such as perifosine and AEZ 108, which targets endometrial, ovarian, prostate and bladder cancer, he thinks there is increasing value in some of the company’s earlier stage drugs such as macimorelin (AEZS-130), a therapeutic treatment for severe chronic diseases such as cancer induced cachexia.
In a research note today Lowe says “Our model currently ascribed greater value to more advanced clinical-stage cancer assets like perifosine and LHRH-doxorubicin conjugate AEZS-108, but macimorelin now has two value-enhancing programs that could impact product revenue in future years.”
Despite a sluggish recent performance on the stock, Lowe said the company’s strong pipeline is reason to keep the faith on AEZ. He maintained his Speculative Buy rating and US$3.50 target price on AEZ based on net present value and “multiples of F2017 forecasts, discounted at 30%.” Loe believes that Aeterna Zentaris’ revenue will be north of $150 million that year.
Shares of AEterna Zentaris on the TSX closed today up 1.2% to $1.70.
Nick Waddell
Founder of Cantech Letter
Cantech Letter founder and editor Nick Waddell has lived in five Canadian provinces and is proud of his country's often overlooked contributions to the world of science and technology. Waddell takes a regular shift on the Canadian media circuit, making appearances on CTV, CBC and BNN, and contributing to publications such as Canadian Business and Business Insider.