Raymond James analyst Rahul Sarugaser updated clients on Thursday on biotech company Eupraxia Pharmaceuticals (Eupraxia Pharmaceuticals Stock Quote, Charts, News, Analysts, Financials TSX:EPRX), saying the appointment of a new Chief Medical Officer raises confidence in the company’s upcoming clinical trial readout.
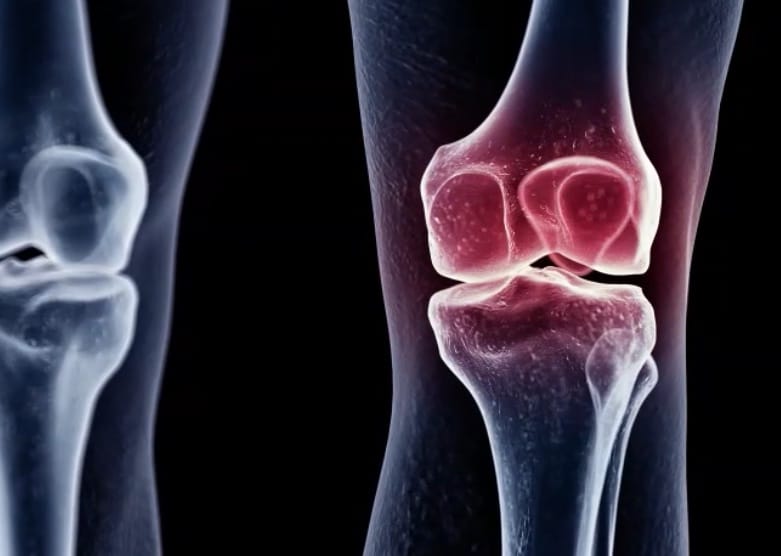
Victoria, BC-based Eupraxia is a clinical stage biotechnology company focused on locally-delivered, extended-release therapeutic candidates, with lead drug P-104IAR in trials for the treatment of osteoarthritis (OA) of the knee and for treatment of eosinophilic esophagitis (EoE).
The company announced on Thursday the appointment of Mark Kowalski, MD, PhD, as its Chief Medical Officer (CMO), effective immediately.
“As we approach our Phase 2 readout with EP-104IAR in osteoarthritis, expected in the second quarter of this year, together with the broadening of our pipeline with additional drug candidates, the need for an experienced CMO has become compelling. We are pleased to attract a candidate of Dr. Kowalski’s calibre to this critical role and welcome Mark to the team,” said CEO Dr. James Helliwell in a press release.
Eupraxia has completed recruitment of its Phase 2 trial in OA and it has maintained its guidance of topline readout from the trial in the second quarter of 2023, with Sarugaser saying the results should arrive by mid-June, with full data likely to have been collected by now.
Sarugaser said he expects management to leverage any data it has collected to date to make strategic decisions about its future, including now the hiring of its new CMO in preparation for a Phase 3 trial.
On the new hire, the analyst wrote, “We read this as a positive leading indicator of strong top-line data from its Ph2 clinical trial in OA, which should read out in a matter of weeks, so we raise our rating on EPRX to Strong Buy, and reiterate our $11 TP.”
Sarugaser said Dr. Kowalski is a highly experienced Phase 3 clinical program-oriented CMO and that Kowalski would have reviewed all available data before deciding to join the company.
“Dr. Kowalski, we expect, would view this data through the lens of a commercially-minded clinician who, it appears, appreciates the demonstrated safety and likely efficacy of the drug. This, in our view, bodes very well for EPRX, and raises our confidence in a positive top-line readout from the Ph2 OA trial in mid-late Jun,” he said.
Sarugaser further added that given the mode of action in OA is identical to that of EoE, a positive read-out from the Phase 2 OA trial would de-risk the read-out of the EoE trial, as well, expected in a couple of months.
“This further consolidates our growing confidence, and undergirds our re-rating to Strong Buy,” he said.
With the new “Strong Buy” rating (previously “Outperform”), Sarugaser’s $11.00 target represented at press time a projected return of 56 per cent.



 Share
Share Tweet
Tweet Share
Share




Comment