Say goodbye to the vasectomy gentlemen, contraceptive gel is here
 Scientists have come up with a male contraceptive gel which has been proven effective in blocking the release of sperm in a study conducted on primates.
Scientists have come up with a male contraceptive gel which has been proven effective in blocking the release of sperm in a study conducted on primates.
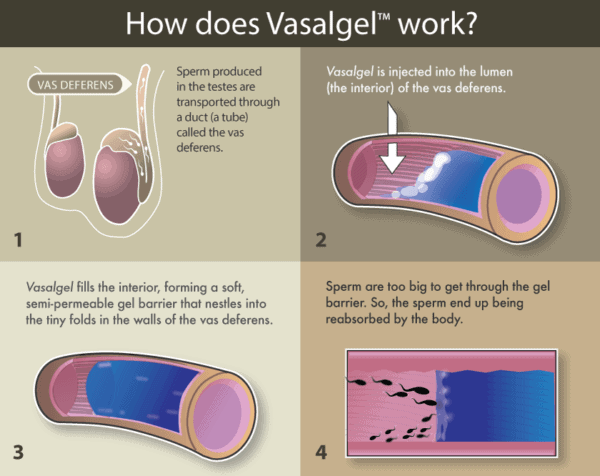
The product is called Vasalgel and the process involves injecting a small amount of the gel into the vas deferens, the tube that carries sperm from the testicles to the urethra. After injection, the gel adheres to the tissue within the vas deferens, effectively creating a barrier which blocks the passage of sperm.
The contraceptive gel is a much simpler and less invasive procedure than the common vasectomy…
Overall a much simpler and less invasive procedure than the common vasectomy, which involves severing the vas deferens and tying or sealing up the two ends, the Vasagel barrier can also be reversed much more easily, by simply flushing out the gel with a sodium bicarbonate solution.
“Men’s options for contraception have not changed much in decades,” says Catherine VandeVoort, lead author of the new study and researcher at the California National Primate Research Centre at the University of California, Davis, to the Guardian. “There’s vasectomy, which is poorly reversible, and condoms. If [men] knew they could get a reliable contraceptive that could also be reversed I think it would be appealing to them.”
Researchers injected the Vasalgel into 16 adult male rhesus monkeys at the Primate Research Center, ten of whom had previous conceptions. The monkeys were housed in large outdoor enclosures with exposure to females rhesus monkeys for at least one full breeding season and in some cases two. No conceptions occurred for any of the monkeys, which showed that in this trial the procedure was effective. A similar procedure using Vasalgel had been tested on rabbits last year with the same results.
Importantly, the research team found no adverse reactions to the implanted gel, as the material did not cause localized inflammation or have any perceived negative reaction with the vas deferens tissue.
“Vasalgel placement into the vas deferens of mature male rhesus macaque monkeys produced reliable contraception, as evidenced by the lack of pregnancies in sexually mature, reproductively viable females with which the study males were co-housed,” say the new study’s authors, whose research is published in the journal Basic and Clinical Andrology.
The makers of Vasalgel are reportedly now starting trials with men.
The news comes just as a hormone-based male contraceptive had its clinical trial put on hold this past November for producing side effects in some of its participants. The study involved long-acting progestogen and testosterone injections given to 320 men between the ages of 18 and 38, with researchers finding that the hormones produced mood swings, depression and acne in a number of cases, causing 30 participants to quit the trial.
“The frequencies of mild to moderate mood disorders were relatively high,” the study read. “The most common adverse events were acne, injection site pain, increased libido, and mood disorders. Following the recommendation of an external safety review committee the recruitment and hormone injections were terminated early.”
Researchers did find that the hormone contraceptive was effective in almost 96 per cent of participants who continued the trial.

