

Waterloo, Ontario’s Intellijoint Surgical Inc. has closed an $11 million Series A round of financing, led by “private investors” who do not wish to be identified from the Waterloo-Toronto corridor, in multiple tranches.
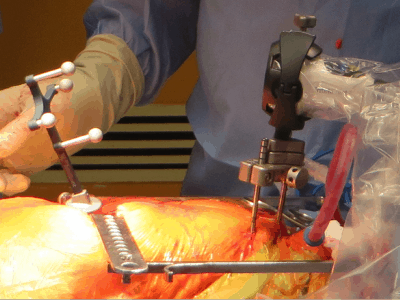
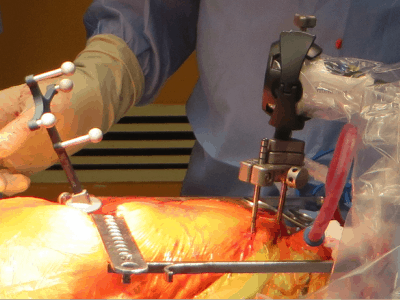
The company’s flagship intellijoint HIP product is an example of miniature 3D surgical measurement technology and mini-optical navigation in the sterile field, a smart surgical instrument that allows orthopaedic surgeons to control several key measurements to ensure a better outcome for hip replacement patients.
“The US launch of the next generation intellijoint HIP earlier this year is being extremely well received in the marketplace and is seeing significant growth,” said Intellijoint Surgical CEO and Co-Founder, Armen Bakirtzian. “This Series A capital will allow Intellijoint to enhance its product offering with the Direct Anterior Approach Application and allow for expansion into new US markets while enabling deeper penetration of Intellijoint’s presence in New York and Illinois.”
intellijoint HIP helps orthopaedic surgeons reach preoperative surgical targets by providing intraoperative measurements in real time, enabling proper establishment of cup position, equalization of leg length and restoration of hip offset during Total Hip Arthroplasty (THA) procedures, which enables surgeons and hospitals to improve surgical outcomes by reducing risk, while improving patient outcomes and lowering cost.
Being able to correct things like hip offset and leg length during surgery should lead to greater efficiency and an end to needless post-surgery recalls, not to mention lawsuits, given that currently 62% of patient legs are lengthened up to 1 cm after surgery, while half of patients will perceive this discrepancy 12 months after surgery, resulting in 30% dissatisfaction five to eight years after the procedure.
Furthermore, up to 78% of patients have been involved in a post-op legal dispute with surgeons, with leg length discrepancy being one of the most frequent complaints.
In March, Intellijoint expanded into the international marketplace, entering into a strategic partnership with orthopaedic innovator Corin Group, giving Corin distribution rights in Australia for Intellijoint’s flagship intellijoint HIP product.
In January, the intellijoint HIP was awarded US Food and Drug Administration clearance.
Founded in 2010, Intellijoint Surgical’s intellijoint HIP has been used in over 500 procedures in the US and Canada, and is both Health Canada licensed and FDA approved.
Intellijoint Surgical is the recipient of the 2015 North American Frost & Sullivan Enabling Technology Leadership Award and the Futurpreneur Shopify True Grit Award 2016.
The company’s R&D team consists of renowned orthopaedic surgeons, including Dr. Allan Gross at Mount Sinai Hospital in Toronto, Dr. Wayne Paprosky in Chicago, Dr. Javad Parvizi at Thomas Jefferson University Hospital in Philadelphia, Dr. Michael Cross at the Hospital for Special Surgery and Ran Schwarzkopf at the NYU School of Medicine in New York.
“Intellijoint has addressed the shortcoming of traditional navigation. The miniature optical camera provides accurate measurements while compensating for patient movement, which is routine during a total hip replacement. It provides me with valuable information and I choose to use it in every case,” added Dr. Paprosky, who is also Midwest Orthopaedics at Rush and member of Intellijoint’s scientific medical advisory board.
Leave a Reply
You must be logged in to post a comment.


 Share
Share Tweet
Tweet Share
Share




Comment