Canadian researchers create army of targeted cancer fighting nanorobots
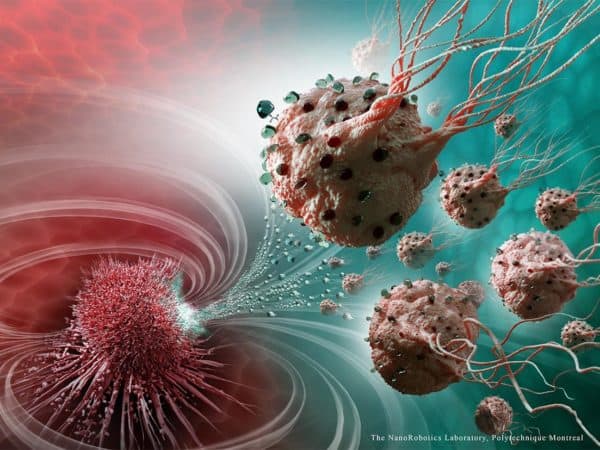
 A team of Canadian researchers have created an army of highly targeted cancer-fighting nanorobots.
A team of Canadian researchers have created an army of highly targeted cancer-fighting nanorobots.
In what’s being hailed as a spectacular breakthrough in cancer research, scientists from Polytechnique Montréal, Université de Montréal and McGill University have created a new breed of nanorobotic agents that are capable of delivering cancer drugs directly to hard-to-reach cancer cells, effectively “throwing the door wide open” to the development of highly targeted forms of cancer treatment.
While cancer research has so far been successful at producing a wide range of chemotherapy drugs capable of attacking and killing cancer cells, one of the main drawbacks to these drugs has been their indiscriminate nature -they often end up killing healthy cells and tissues along with the cancerous ones, damaging a patient’s organs and taxing the immune system.
The issue is especially problematic when dealing with the oxygen-depleted areas within cancer tumors, called hypoxic regions, which cannot be readily accessed by pharmaceutical agents that need to travel via the body’s bloodstream. In fact, studies have shown that for current therapy options using drug-carrying molecular compounds – natural “nanocarriers” such as liposomes and micelles – only a small fraction (roughly two per cent) of the administered drugs actually reach the hypoxic regions in tumors.
But in a new study published in the journal Nature Nanotechnology, researchers have figured out a way to use a strain of Magnetococcus marinus bacteria known as MC-1 as their drug-delivering agent, capable of specifically targeting cells in hypoxic areas. The key lies in MC-1’s geomagnetics – each bacterial cell contains a chain of magnetic iron-oxide nanocrystals which enable it to move directionally within a magnetic field, in this case, allowing it to travel towards areas of low oxygen concentration.
“These legions of nanorobotic agents were actually composed of more than 100 million flagellated bacteria – and therefore self-propelled – and loaded with drugs that moved by taking the most direct path between the drug’s injection point and the area of the body to cure,” says Professor Sylvain Martel, Canada Research Chair in Medical Nanorobotics at Polytechnique Montréal and co-author of the study.
Working with mice afflicted with colorectal tumors, the researchers were able to successfully administer their MC-1 nanocarriers right to the targeted areas, with upwards of 55 per cent of MC-1 cells penetrating into hypoxic regions of the mice tumors. “The drug’s propelling force was enough to travel efficiently and enter deep inside the tumors,” says Professor Martel.
The research team sees their finding to have potential uses not only in the targeted delivery of cancer drugs but in other approaches to treatment such as radiotherapy which could benefit from a similar guided delivery model. “This innovative use of nanotransporters … throws the door wide open to the synthesis of new vehicles for therapeutic, imaging and diagnostic agents,” says Professor Martel. “Chemotherapy, which is so toxic for the entire human body, could make use of these natural nanorobots to move drugs directly to the targeted area, eliminating the harmful side effects while also boosting its therapeutic effectiveness.”

