Prometic Life Sciences should have strong clinical news flow in 2015


This past Friday, the tape was telling investors that something was up at Prometic Life Sciences (Prometic Stock Quote, Chart, News: TSX:PLI).
Prometic stock was surging on above average volume, even though there was no news from the company. Ultimately, IIROC stepped in and halted the stock late in the day due to pending news.
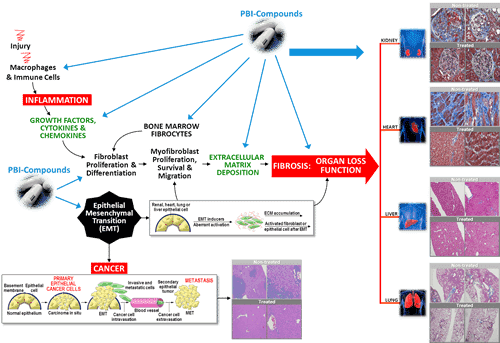
Yesterday, investors learned that the cause for all the commotion in Prometic trading was that the company had received orphan drug designation from FDA for its drug PBI-4050 in the treatment of idiopathic pulmonary fibrosis (IPF).
The news about the orphan designation is a clear positive for Prometic as it pushes ahead with PBI-4050 in IPF. Having the orphan designation offers Prometic both potential pre-approval and post-approval benefits, including tax credits, reduction or elimination of certain FDA administration fees, and most importantly seven years market exclusivity if PBI-4050 were to be approved.
Of course, in order to receive any of these potential FDA benefits Prometic needs to first demonstrate the efficacy of PBI-4050 for IPF in man. To date, the company has only completed animal studies in IPF, and although the data has been compelling, the step into human efficacy studies will be the true test.
Prometic earlier this month announced it had received Health Canada clearance to commence a Phase II IPF study with PBI-4050. The company is planning a 12 study, so investors shouldn’t have to wait too long to get an initial sense of how PBI-4050 performs in IPF patients.
Not only will the company be starting a Phase II IPF study shortly, it received Health Canada clearance to start another Phase II with PBI-4050 for the treatment of patients with metabolic syndrome (and associated type II diabetes). Prometic has also completed enrollment in a Phase IB study in patients with diabetic kidney disease, with data expected this quarter.
There should be no shortage of clinical news flow and data from Prometic in 2015 – consider yourself duly notified.

Hogan Mullally
Writer