The stock remains well off its highs, but investors will want to be thinking of Mind Medicine (Mind Medicine Stock Quote, Charts, News, Analysts, Financials NASDAQ:MNMD), according to Roth Capital Partners analyst Jonathan Aschoff, who reported on the biopharma company on Wednesday and reiterated a “Buy” rating.
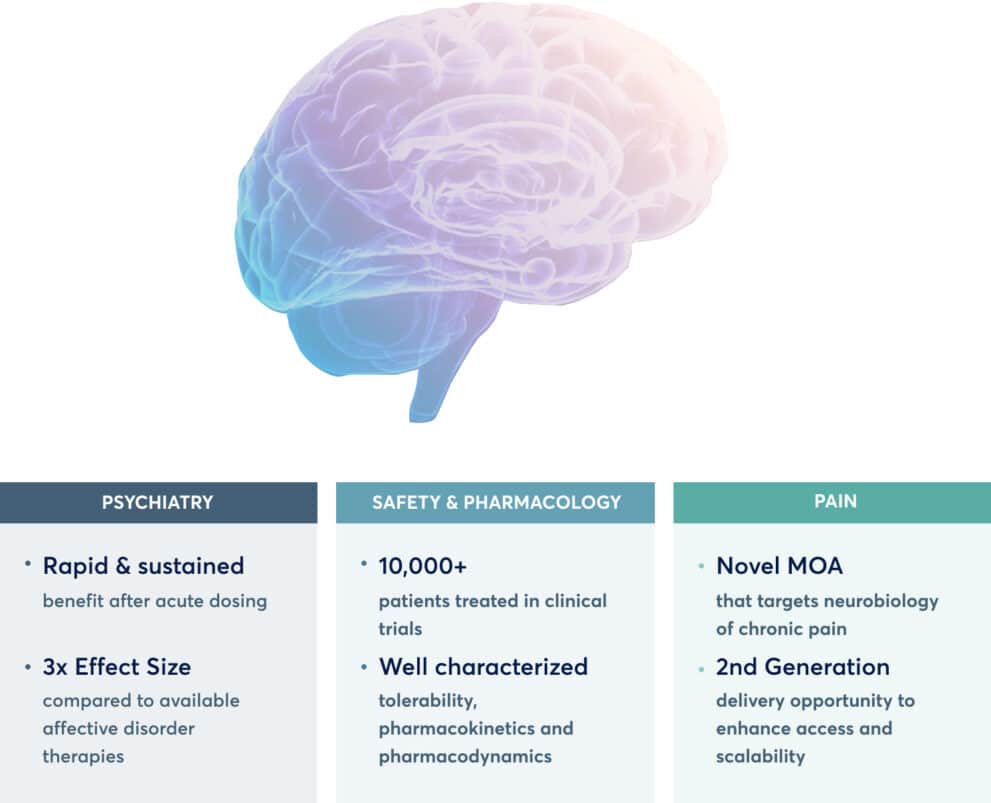
Mind Medicine is a clinical-stage developer of candidates to treat brain health disorders, with a pipeline of assets targeting neurotransmitter pathways playing a role in brain health disorders. The company held on Monday an Investor Day, where it provided an update on MM-120 (lysergide D-tartrate), in trials for generalized anxiety disorder (GAD).
MindMed said the current Phase 2b trial for MM-120 remains on track for a late 2023 topline data readout. The trial has participants receiving a single administration of the drug at various dose strengths, with the aim being to measure reduction in anxiety symptoms four weeks after the single administration. Secondary objectives will be measuring results up to 12 weeks after dosing.
MM-120 is a pharmacologically optimized form of LSD being developed for GAD and other brain health disorders. Mind Medicine received clearance from the US FDA for its Investigational New Drug (IND) application in January 2022, allowing it to proceed with the Phase 2b trial.
Aschoff commented on the discussions from four experts who spoke at the Investor Day, starting with Dr. Maria Oquendo, who said anxiety, depression and alcohol use disorder are indications in which LSD has seen clinical success, which somewhat reduces the clinical risk for MM-120, in Aschoff’s view, at least for the GAD trial to read out by the year end. Dr. Oquendo put the US GAD prevalence at about 2.9 per cent of the population, 76 per cent of which have a moderate to severe condition that disrupts th patient’s ability to function effectively either socially or at work.
Next, Dr. David Feifel said that one in five people in the US takes psychiatric drugs and that the crisis is the low number of new drugs in the space. Dr. Feifel’s view of using psychedelics for treating disease is highly favourable compared to his view of SSRIs for many of the same conditions and that the large increase in behavioural health conditions in recent years has pushed the FDA to be more permissive than ever in allowing clinical trials with what were once viewed as purely recreational drugs.
Aschoff said CEO Rob Barrow’s closing remarks included comments on MM-120’s rapid and durable effect and how 65 per cent of patients taking LSD in other trials had clinical benefit versus nine per cent of placebo.
“We look forward to Phase 2b and 2a MM-120 data in GAD and ADHD, respectively by YE23, followed by an EOP2 meeting that should nail down a Phase 3 plan,” Aschoff wrote.
With the update, Aschoff maintained a 12-month target on MNMD of $25.00, which at press time represented a projected return of 593 per cent.




 Share
Share Tweet
Tweet Share
Share




Comment