 Tailor made drug molecules are almost here thanks to supercomputers.
Tailor made drug molecules are almost here thanks to supercomputers.
Personalized or precision medicine is changing the way health care will be carried out, with the development of tailor-made drugs being at the forefront of that advance.
And as new research from the University of Washington shows, having access to the data-crunching and modelling power of supercomputers is proving key to the development of this new class of drugs.
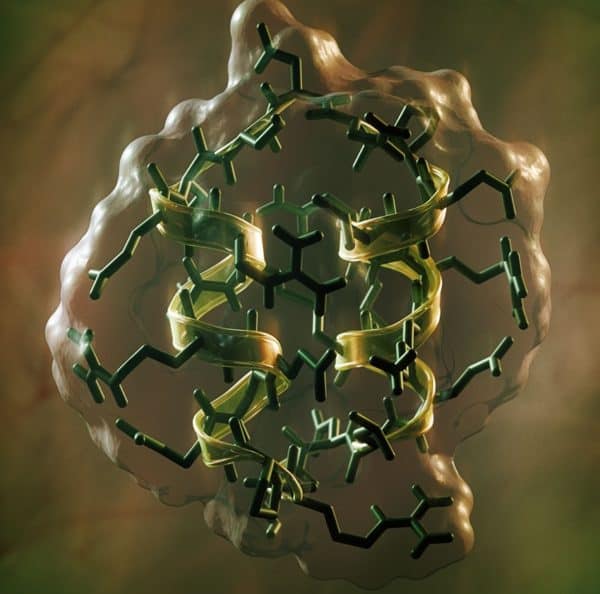
In a new publication in the journal Nature, researchers show how they created stable, synthetic peptides, short chains of amino acids that come together to form more complex proteins. In this case, the peptides are designed so that they’ll bind with and target particular receptor molecules, which makes them well-honed weapons for use in precision medicine, the study’s authors say.
“That you can design molecules from scratch that fold up into structures, some of which are quite unlike what you see in nature, demonstrates a pretty fundamental understanding of what goes on at the molecular level,” said David Baker, a University of Washington biophysicist and lead researcher for the new study, in a statement. “That’s certainly one of the more exciting things about this work.”
In order to create their peptides, the research team employed the vast computing power of the US Department of Energy’s Mira supercomputer at its Argonne Leadership Computing Facility in Lemont, Illinois. The computer modelling sampled millions upon millions of possible shapes for amino acids to find ones that could be rendered stable and, eventually, could be applied in therapeutic use.
The computer modelling was followed up with lab replication of the chosen configurations, which turned out to confirm the stability of the synthetic structures.
“At best, what comes out of a computer is a prediction, and at worst what comes out of a computer is a fantasy. So we never really consider it a result until we’ve actually made the molecule in the wet lab and confirmed that it actually has the structure that we designed it to have,” said Vikram Mulligan, research at the University of Washington and study co-author.
Tailor made drug molecules: the advantage is in the targeting…
The real advantage of these proposed designer drugs is that unlike conventional drugs, which are more widespread in terms of the kinds of molecules in the body to which they may bind (in some cases, producing unwanted side effects), peptide-based drugs have very specific target binding agents, making them both more effective and less prone to side effects.
More focused therapeutic medicines are being created to fight cancer by attacking specific proteins that play a role in many cancers. At the University of Toronto’s Centre for Medicinal Chemistry, for example, U of T professor Patrick Gunning says that he and his team are working on tailored molecules to attack STAT3 and STAT5 proteins in the body without harming surrounding healthy cells.
“General chemotherapy and radiation tend not to be selective,” says Dr. Gunning in a U of T press release. “As a result, you have general toxicity in the human body. With targeted therapeutics, you hit fewer things and . . . it’s a much softer treatment.”
“We’re almost there,” says Dr. Gunning. “Our molecules have killed cancer cells in numerous pre-clinical trials. Now we need to get them into clinical trials, so we can start saving lives.”
Leave a Reply
You must be logged in to post a comment.




 Share
Share Tweet
Tweet Share
Share



Comment