 The US Federal Drug Administration has announced approval of the first drug used specifically to treat giant cell arteritis, an autoimmune disorder that causes inflammation of the blood vessels of the head.
The US Federal Drug Administration has announced approval of the first drug used specifically to treat giant cell arteritis, an autoimmune disorder that causes inflammation of the blood vessels of the head.
The drug tocilizumab, manufactured by Swiss multinational Roche under the name Actemra, is already in use as an immunosuppressive mainly to treat rheumatoid arthritis. The approved expansion stems from a double-blind, placebo-controlled study involving 251 patients with giant cell arteritis, which found that patients given subcutaneously administered Actemra were more likely to achieve sustained remission of symptoms than those who did not take the drug. Study participants on Actemra were also able to lower their use of prednisone, a corticosteroid commonly used to treat giant cell arteritis.
“We expedited the development and review of this application because this drug fulfills a critical need for patients with this serious disease who had limited treatment options,” said Badrul Chowdhury, M.D., Ph.D., director of the Division of Pulmonary, Allergy, and Rheumatology Products in the FDA’s Center for Drug Evaluation and Research, in a press release.
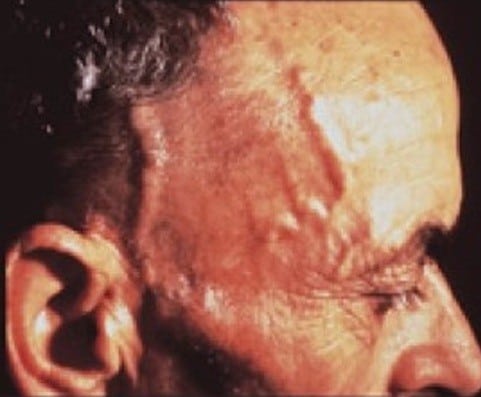
Like other autoimmune diseases, giant cell arteritis occurs when the body’s immune system starts attacking itself, in this case, causing inflammation of the medium- and large-sized arteries. Most often occurring in the head but also in the chest, neck and other areas, the inflammation causes a narrowing of the arteries and restriction in blood flow, producing symptoms such as vision problems, headache, weak or painful jaw muscles, fatigue and weight loss. Untreated, giant cell arteritis can lead to blindness, stroke, heart attack and other conditions caused by impaired blood flow.
Corticosteroids can be effective at controlling symptoms but some patients need to keep taking them for one to five years or even indefinitely to keep the condition under control. Giant cell arteritis is most common in the elderly and rarely occurs in people below the age of 50, developing in women more often than men and Caucasians more often than other groups.
The FDA states that Actemra will carry a boxed label warning of the possibility of serious infections, that live vaccines should be avoided during treatment with Actemra and that the drug should be used with caution among people with increased risk of gastrointestinal perforation or dangerous allergic-like hypersensitivity reactions.
“Today’s FDA decision means people living with giant cell arteritis will, for the first time, have an FDA-approved treatment option for this debilitating disease. With no new treatments in more than 50 years, this approval could be transformational for people with GCA and for their physicians,” said Sandra Horning, chief medical officer and head of Global Product Development, Genentech, a Roche subsidiary.
A relatively rare disorder, giant cell arteritis affects an estimated 200 out of 100,000 people over the age of 50. A 2016 Canadian study found that the survival rate for patients with giant cell arteritis improved dramatically over the past 20 years.
Funded by the Canadian Institutes of Health Research, the study looked at all newly diagnosed cases of the disease occurring in British Columbia between the years 1997 and 2012 and found that the early cohort of patients between 1997 and 2004 had a much higher mortality rate of 373.7 cases per 1,000 person-years in comparison with the later cohort between 2005 and 2012, where the mortality rate was 87.5 cases per 1,000 person-years.
Leave a Reply
You must be logged in to post a comment.





 Share
Share Tweet
Tweet Share
Share




Comment