 A major milestone for Oncolytics Biotech (Oncolytics Biotech Stock Quote, Chart, News: TSX:ONC) is being cheered by Canaccord Genuity analyst Neil Maruoka.
A major milestone for Oncolytics Biotech (Oncolytics Biotech Stock Quote, Chart, News: TSX:ONC) is being cheered by Canaccord Genuity analyst Neil Maruoka.
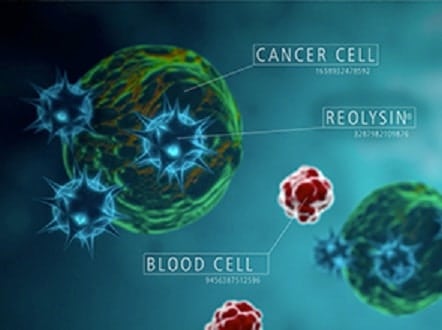
On Friday, Oncolytics Biotech announced positive data from its 74-patient, phase 2 Reolysin study. The study assessed the therapeutic combination of intravenously administered Reolysin given in combination with paclitaxel versus paclitaxel alone in patients with advanced or metastatic breast cancer.
“There is an emerging pattern, from this and other studies with Reolysin, where patients obtain significant benefit in overall survival, despite limited impact on response rates and/or progression-free survival,” said Oncolytics Chief Medical Officer Dr. Andres Gutierrez. “This is a well-established pattern for other immunotherapies, like checkpoint inhibitors, which have been approved on an overall survival primary end point in melanoma, [non-small-cell lung cancer], and head and neck cancers. These phase 2 data also support the established mode of activity of Reolysin where selective cell lysis of permissive cancer cells is followed by an anti-tumour immune response, which may be responsible for the meaningful survival benefit for patients. Taking into account the specific findings from this study, we continue to believe that Reolysin is not solely an oncolytic agent but has key attributes of an immuno-oncology agent as well.”
Maruoka says Friday’s news is an unambiguous positive for Oncolytics Biotech.
“We have long been of the view that Reolysin is efficacious; however, this has been difficult to demonstrate in a randomized clinical trial,” he says. “We believe the reason is that, as an immunotherapeutic agent, Reolysin is less likely to positively impact the endpoints of Progression-Free Survival (PFS) and Response Rate (RR), but is more apt to result in an improvement in Overall Survival (OS). OS is a more difficult endpoint to measure (as it often takes longer to evaluate), but is a more important and relevant endpoint, in our view. Patients receiving Reolysin with standard of care showed a 67.3% improvement in OS; not surprisingly, patients failed to show a meaningful improvement on the primary endpoint of PFS or RR. Regardless, we believe this is an excellent result for Reolysin, validating the survival data we have seen so far from the Phase II pancreatic cancer study.”
In a research update to clients today, Maruoka maintained his “Buy” rating and one-year price target of $1.00 on Oncolytics Biotech, implying a return of 66.7 per cent at the time of publication.
Maruoka thinks Oncolytics Biotech will generate EBITDA of negative $24.5-million on revenue of zero in fiscal 2017.
Comment
One thought on “Oncolytics Biotech is undervalued, says Canaccord Genuity”
Leave a Reply
You must be logged in to post a comment.




 Share
Share Tweet
Tweet Share
Share




You are going to have to run to keep up with ONCY this year. $1 is just the beginning, more good news in the offing.