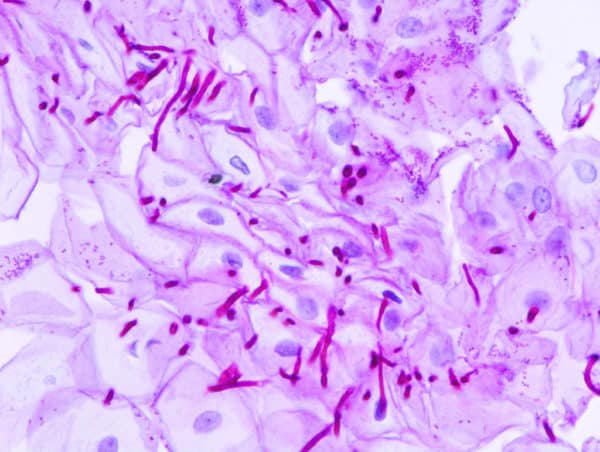

A micrograph of esophageal candidiasis showing hyphae, biopsy specimen, PAS stain.
New Canadian research may mean a breakthrough in the fight against a deadly fungus and could further our understanding of how microbial communities affect human health.
The University of Toronto study, called “Global Analysis of the Fungal Microbiome in Cystic Fibrosis Patients Reveals Loss of Function of the Transcriptional Repressor Nrg1 as a Mechanism of Pathogen Adaptation” was published Friday in the journal PLoS Pathogens. Researchers looked at phenotypic and genotypic analyses of fungi from 89 sputum samples sourced from 28 cystic fibrosis patients.
Various fungi are often recovered from cystic fibrosis patient sputum samples and are thought to be linked to the deterioration of lung function, but the study’s researchers say our understanding of them is still “in its infancy”. In particular, the researchers were interested in how fungi change with the progression of disease or with treatment.
“Fungi have a staggering impact on human health, infecting billions of people around the world and killing 1.5 million every year – that’s in the range of tuberculosis and malaria.” And yet, they are underappreciated and not well understood,” said Leah Cowen, who was the lead researcher on the study, and is a U of T Molecular Genetics professor and also Canada Research Chair in Microbial Genomics and Infectious Disease.
Researchers were unsurprised to find the presence of pathogenic fungi Candida and Aspergillus, but were surprised to find an interaction between certain fungi and bacteria.
“Some of this fungi began shifting into its stringy shape without any environmental cue – usually this transformation (called filamentation) doesn’t happen spontaneously, but is triggered by the presence of certain substances, such as blood,” notes Eureka Alert. “To see if there could be a genetic explanation, the researchers sequenced the genomes of these samples and found a common denominator. All but one had genetic mutations in a gene known to repress the change shape – called NRG1.”
“This was a smoking gun,” explains Cowen. “This gene makes a protein that stops filamentation – like a brake. Because of these genetic mutations, the fungi lost this brake and were not able to stop these long strings from forming.”
Fungal infections kill an approximately 1.3-million people every year. One expert thinks a general lack of public awareness of them is partly to blame.
“People know well what a bacterium or a virus can do,” says David Perlin, executive director of the Public Health Research Institute at Rutgers New Jersey Medical School. “We need to start thinking of fungi in the same terms, and part of that is becoming more aware that fungi are all around us in the environment. Yeast is a fungus. So is mold. Many people don’t realize that.”
Perlin notes that more than a million people worldwide are blind due to fungal infections, and 350,000 asthma -related deaths each year are linked to fungus.
The U.S. Centers for Disease Control and Prevention is currently tracking multiple fungal outbreaks and investigations, including cases of Coccidioidomycosis (Valley fever) in California Prisons, Histoplasmosis in an Illinois Prison, and an outbreak of Outbreak of mucormycosis after a tornado hit Joplin, Missouri.
Leave a Reply
You must be logged in to post a comment.




 Share
Share Tweet
Tweet Share
Share




Comment