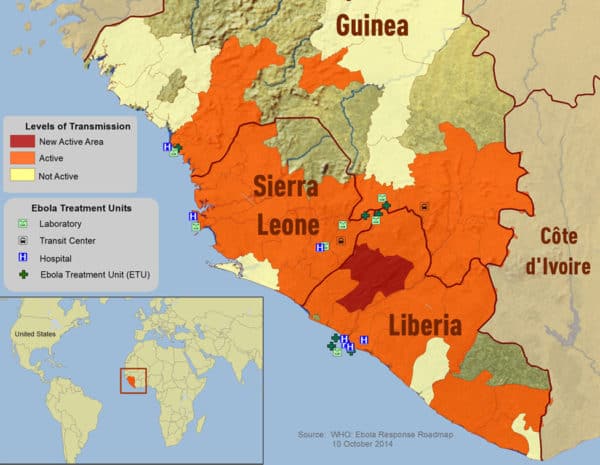
 News report: Canadian Ebola vaccine to be shipped to Geneva next week.
News report: Canadian Ebola vaccine to be shipped to Geneva next week.
Along with the rest of our planet, I dearly hope that the researchers at Winnipeg’s National Microbiology Laboratory have discovered the 21st century’s version of Major Sir Frederick Banting, MC’s insulin discovery (and then some).
What will come as a surprise, perhaps, is the news that the Public Health Agency of Canada has exclusively licenced the intellectual property, and not to a Canadian biotech firm. As reported by The Globe and Mail:
Canada holds the intellectual property rights to the vaccine but has licensed the rights to a small American biotech company, NewLink Genetics. Based in Ames, Iowa, the company’s primary focus — until recently — has been the development of cancer vaccines. It does not have its own vaccine production facility and has never brought a product through the expensive and onerous process of gaining regulatory approval.
But because it holds the licence to one of a very few experimental Ebola vaccines — and one of only two ready for human safety trials — NewLink has found itself at the centre of a storm.
While the company has been getting assistance from a U.S. government agency — the Biomedical Advanced Research and Development Authority, or BARDA — frustrated scientists and others have questioned whether the company has the resources, finances and clout to push the vaccine forward.
NewLink Genetics (NewLink Genetics Stock Quote, Chart, News: NLNK:Q) listed on the NASDAQ three years ago on the back of work its researchers had done to use immunotherapies to treat cancer. Like most early stage biotechs, the company is still looking for meaningful revenue from that research. To date, NewLink has burned more than US$150 million, and had cash on hand of about US$78 million as of Q2 (or 8 quarters of cash at current burn rates).
Click here to finish reading this article.
Leave a Reply
You must be logged in to post a comment.





 Share
Share Tweet
Tweet Share
Share




Comment