 In what researchers are calling a “simple and elegant” solution to the problem of what to do with all that carbon dioxide, scientists at the University of Toronto have discovered a conversion process for creating energy from CO2 using two very abundant and very cheap materials -sand and sunlight.
In what researchers are calling a “simple and elegant” solution to the problem of what to do with all that carbon dioxide, scientists at the University of Toronto have discovered a conversion process for creating energy from CO2 using two very abundant and very cheap materials -sand and sunlight.
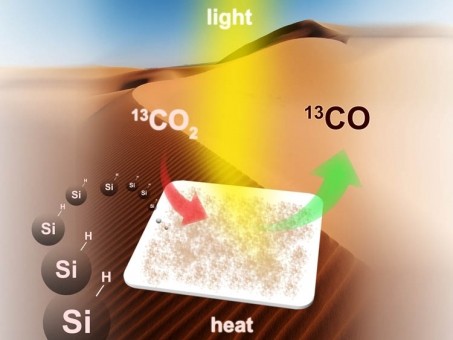
Measuring just 35 nanometers in size, silicon nanocrystals have unique electrical and optical properties that allow it to absorb near-infrared, visible and ultraviolet light, thus making it an excellent candidate for converting carbon dioxide into carbon monoxide, a usable hydrocarbon fuel.
“It is indeed a surprising yet welcome discovery that plentiful, inexpensive and benign elemental silicon, synthesized in the form of ncSi:H made easily and cheaply from commercially available SiO, can exploit the reducing power of surface hydride to chemically reduce CO2 to CO,” say the new study’s authors whose research is published in the journal Nature Communications.
The discovery is part of an emerging field of solar fuels research which is investigating various methods for capturing the energy emitted from the sun by converting it into chemical energy – in this case, by reducing carbon dioxide to carbon monoxide which can be more readily translated into a fuel source.
But so far, a major stumbling block has been the need to add rare and expensive elements to serve as catalysts or semiconductors for the conversion process. That problem may now be behind us, say the study’s authors, members of the University of Toronto’s Solar Fuels Cluster, an interdisciplinary research team investigating the energy harvesting capabilities of CO2.
Researchers studied the reactivity of silicon nanocrystals when exposed to a metal halide lamp, both in the absence and in the presence of hydrogen gas at 150 degrees celsius. The results showed conclusively that the silicon crystals can reduce CO2 to CO.
“In the war against climate change, the consensus is that carbon dioxide capture and storage alone cannot solve this problem and to have a meaningful effect needs to be combined with chemical and catalytic processes that convert the carbon dioxide into value-added chemicals and fuels,” say the study’s authors. “It might well take something as simple and elegant as CO2 fixation by ncSi:H to solve such a monumental global problem.”
The research comes on the heels of another recent solar fuel discovery at the University of Illinois at Chicago which proved that the conversion of carbon dioxide into useful energy can be accomplished with a solar cell that uses compounds called transition metal dichalcogenides,or TMDCs, as catalysts for the solar-powered reduction of CO2. The resulting photosynthetic process produces syngas, or synthesis gas, a mixture of hydrogen gas and carbon monoxide which can be either be burned by itself or further converted to a hydrocarbon fuel.
“The new solar cell is not photovoltaic – it’s photosynthetic,” says Amin Salehi-Khojin, assistant professor of mechanical and industrial engineering at UIC and senior author of the study. “Instead of producing energy in an unsustainable one-way route from fossil fuels to greenhouse gas, we can now reverse the process and recycle atmospheric carbon into fuel using sunlight.”
Comment
One thought on “U of T scientists discover new method for creating energy from CO2”
Leave a Reply
You must be logged in to post a comment.



 Share
Share Tweet
Tweet Share
Share




Sure replace harmless CO2 with deadly CO. What kind of moronic research is this?