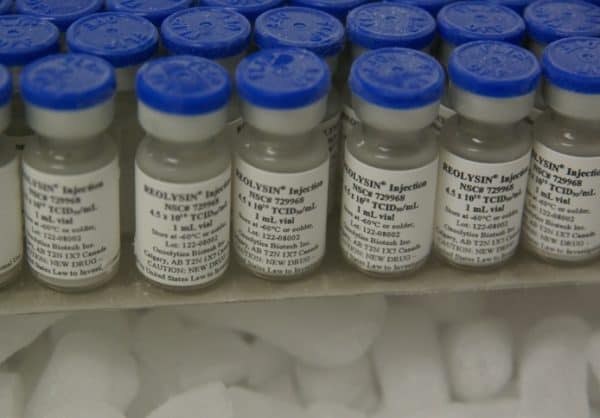
 Shares of Oncolytics Biotech (TSX:ONC) are soaring today after the company announced that a Phase Two study showed its Reolysin treatment was 95% effective in treating tumours.
Shares of Oncolytics Biotech (TSX:ONC) are soaring today after the company announced that a Phase Two study showed its Reolysin treatment was 95% effective in treating tumours.
The trial consisted of 20 patients who had squamous cell carcinoma of the lung. They were given intravenous administration of Reolysin in combination with carboplatin and paclitaxel. 19 of them exhibited overall tumour shrinkage.
Oncolytics Biotech CEO Dr. Brad Thompson said the results were more than encouraging.
“It’s exciting to have 95 per cent of patients in this study exhibit tumour shrinkage, and these results further suggest that Reolysin may have potential use in neoadjuvant (presurgical) settings,” he said. “Based on these findings, we intend to continue to look at Reolysin as a treatment for cancers of the lung and cancers that metastasize to the lung.”
Oncolytics Biotech’s history goes all the way back to the early 1990′s and discoveries made in the Department of Microbiology and Infectious Diseases at the University of Calgary. The company has since poured ten of millions of dollars into the development of a cancer treatment based around a formulation of reovirus, a family of viruses that can affect the gastrointestinal system and have shown to have oncolytic, or cancer killing properties.
Lung Cancer accounts for 27% of all cancer deaths in the United States. The American Cancer Society estimates there were 159,480 deaths from lung cancer last year, meaning that more people died from lung cancer than colon, breast and prostate cancers combined.
At press time shares of Oncolytics Biotech were up 23.8% to $4.42.
Leave a Reply
You must be logged in to post a comment.




 Share
Share Tweet
Tweet Share
Share




Comment